Trans Fat has been in the news a lot lately here in the US, since the FDA has proposed to ban it from food. Yet hardly anybody seems to know what trans fat is. Fox News host Tucker Carlson responded to the news by saying “Let me be the only person in America who stands up for trans fats by asking the obvious question, which is ‘If they’re so bad for you, why are they in foods?’ And I suspect the answer is probably because they’re delicious.”
No doubt to Carlson’s chagrin, it was Stephen Colbert who immediately rushed to his defence: “Now that might sound like an idiotic thing to say, but let me be the only person standing up for Tucker Carlson: if he’s so stupid, why is he on Fox News?”
Just making up nonsense when you don’t know the answer to your own question does, indeed, sound pretty idiotic. But the question is completely valid, and while Colbert correctly pointed out that trans fats have “no particular taste”, he never actually answered it either. Why are trans fats really in foods?
The short answer is because they’re cheap.
The long answer is quite a bit more interesting, though. It’s a tale of good intentions, scientific breakthroughs, hubris, and a bit of organic chemistry. (The presence of the latter explains why I am writing this blog post instead of Lauren—a combination of an incompetent high school Chemistry teacher and a college course in Organic Chemistry that doubled as a weed-out class for pre-med students has left her with a phobia of the subject.)
Organic Chemistry 101
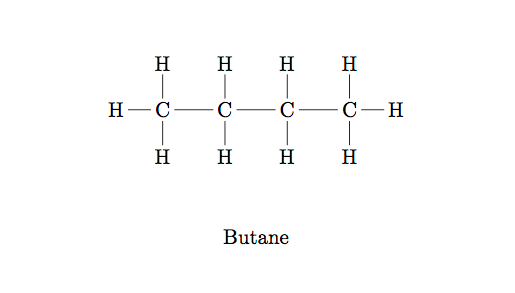
Both fats and carbohydrates (two of the three macronutrients that are essential to animal diets) are comprised of various arrangements of hydrocarbons—that is, chains of Carbon atoms, surrounded by Hydrogen atoms, in various configurations. Each Carbon atom has four available bonds, each Hydrogen atom one, and the bonds repel each other. Other elements can be incorporated in the chain, and have varying numbers of available bonds. There’s an enormous variety of different combinations, but they all stem from a small set of building blocks so their names are derived in a mechanical way according to a fairly simple grammar. The simplest hydrocarbons are called alkanes, so let’s look at one of those first:
Hydrocarbons are named for the lengths of their main chains of Carbon atoms. The first few prefixes are meth-, eth-, prop-, but-, pent-, hex-, … &c. The molecule in the diagram above is an alkane with four Carbon atoms, so it is called butane. Note that this is not a fat—you know it as lighter fluid. If it were a fat, we might say that it is saturated, because it has as many Hydrogens as can possibly fit on a chain of this configuration.
That need not be the case, however. If we take a pair of Hydrogen atoms out, we can form a double bond between two Carbon atoms. For fats, this is the difference between a saturated fat and an unsaturated fat. Simple chains that contain at least one double bond are called alkenes. There are obviously a bunch of ways to do this, which get reflected in the name. There’s usually a number to indicate which of the bonds is the double bond, so if we were to create an alkene (which is still not a fat, by the way) with four Carbon atoms where the second C-C bond is a double, it would be called but-2-ene. But double bonds also have a new property: unlike single bonds, they can’t rotate so any asymmetries are locked in. So, in fact, there are two variants of but-2-ene:
We call these variants cis-but-2-ene (where the two adjacent carbons are on the same side of the double bond) and trans-but-2-ene (where they are on opposite sides), respectively. Bear in mind that this is a 2-dimensional representation of what is really a 3-dimensional structure, but even so you can see that these molecules are quite different despite comprising the same components in the same order.
We haven’t seen a fat molecule yet, but we now already know what a trans fat is: a fat molecule with at least one double bond where the chain continues on different sides of the bond. Immediately you should be suspicious of the entire premise of the trans fat debate. Fats are much more complicated structures than alkanes, and each has many different features in their structure. It’s highly questionable, if not actually absurd, to talk about ‘Trans Fats’ as a single thing, as if the direction of one particular bond does more to determine the behaviour of a molecule than all of its other properties combined. We have located the origin of the name ‘Trans Fat’, only to find that it is shorthand for something else. That something else is the industrial process of partial hydrogenation.
All about Fat
So far we’ve learned the basics of hydrocarbon chemistry with reference to lighter fluid and an industrial ingredient of synthetic rubber that you’ll never encounter. What does a fat look like in comparison? The building blocks are pretty similar. “Fatty acids” are just longer chains with a few Oxygen atoms included in strategic locations. The most common fats in food, known as triglycerides, comprise three of these fatty acid chains joined together at one end. Here’s a typical example:
This triglyceride comprises the fatty acids Stearic Acid (top right), Oleic Acid (left) and Linoleic Acid (bottom right), all attached to a glycerol molecule in the centre. (No chemist actually draws molecules this way, for obvious reasons, but you get the idea. Remember that the real molecule is three-dimensional and all of the single bonds are free to rotate.) Note that Oleic and Linoleic Acids contain one and two cis double bonds, respectively.
By the way, the terms ‘fat’ and ‘oil’ are interchangeable here for our purposes. Fats that are liquid at room temperature are generally known as oils. There are exceptions, but not based on any physical differences. Animal fats contain a high proportion of saturated fat—remember, those are fats with no double bonds. A few plant oils, like coconut and avocado, are saturated fats too. Saturated fats are usually solid at room temperature, thanks to their regular structure (which makes for stronger attraction between molecules).
Most plant oils, however, are polyunsaturated. That means that each molecule includes multiple double bonds. This gives them a much more irregular shape, and as a result polyunsaturated oils are generally liquid at room temperature.
Although polyunsaturated fats are generally considered the best for you, there’s one big down side to them that partially explains why trans fats are still with us: they go rancid much faster than saturated fats. The reason is that rancidity occurs when free radicals break bonds in the fat, initiating an oxidation reaction. The easiest bonds to break are the double bonds. Therefore the more double bonds a fat contains, the sooner it is likely to go rancid.
It is possible to add Hydrogen atoms to an unsaturated fat and turn it into a saturated fat, in a process called (unsurprisingly) hydrogenation. Typically, however, one double bond is left intact to make a monounsaturated fat. Monounsaturated fats are generally semi-solid at room temperature. As you may have guessed already, this process of partial hydrogenation is how margarine is made (and the reason it is softer than butter, which is largely saturated fat).
At the time this was invented, it would have been considered a great achievement of modern science. The prevailing scientific model held that fat was bad for you, and that saturated fat in particular was especially bad for you. Partially hydrogenated vegetable oil could be produced from plants at much lower cost than animal fats—which require huge energy inputs in comparison—plus they lasted longer, were supposedly better for you, and came in a handy spreadable form just in time for the invention of the refrigerator. Everybody wins, right? As we’ll see, this is where the hubris came in.
The process of partial hydrogenation is relatively straightforward in principle (though in practice actual formulations are closely-guarded secrets). Molecules always tend towards their lowest-energy state (that is, if they can release some energy, they will). Imagine you’re on a bicycle in a deep valley—all things being equal, you’re going to eventually end up at the bottom, where you have the lowest energy. A saturated bond has lower energy than a double bond, but before it can form the double bond must first be broken, which requires an input of energy. If there’s a hill between you and the valley, you’re not going to get in there at all unless you add some energy (by pedalling), or magically teleport yourself to the other side of the hill without passing through the space in between. (If you’re laughing, stop, because these are both completely valid options in the world of quantum mechanics, though not really relevant here.) So partial hydrogenation is performed by adding some energy, in the form of heat, plus a catalyst (imagine someone in a bulldozer who will temporarily lower the height of the hill for you).
Here’s the catch: a trans double bond is also a lower energy state than a cis double bond. As a result, the exact same conditions under which most of the double bonds are hydrogenated also cause the remaining double bonds to flip from their naturally-occurring cis configuration to a trans configuration a very high proportion of the time. Trans fat contamination is thus an inevitable result of partial hydrogenation.
There are a few trans fats that occur naturally in our food. For example, a small percentage of milk fat is a polyunsaturated fat with one cis and one trans double bond. (The FDA uses a technical definition of trans fat that excludes these naturally-occurring fats.) Current studies differ on whether this particular fat is bad for you, or actually good for you. However, the trans fats produced by partial hydrogenation are not found anywhere in nature. They are a completely new addition to the human diet in the past century. It turns out that not only is the hypothesis that saturated fat is bad for you on increasingly shaky ground, it is now widely accepted that the monounsaturated trans fats produced by partial hydrogenation of vegetable oils are definitely bad for you, and certainly worse than the saturated fats they were designed to replace.
What to do?
So, this miracle product that was scientifically formulated to make us healthier has actually been making us unhealthier all along. What should we do about that? Compulsory labelling of trans fats on nutrition labels has already done a lot to reduce the amount of trans fat in our diets. As a result of labelling, margarine as we knew it has virtually disappeared from supermarket shelves. In its place are ‘spreads’ mostly made from butter cut with vegetable oil, containing no trans fats. Consumers are clearly embracing the health claims of butter over those of partially-hydrogenated vegetable oils. Even the world’s largest margarine producer, whose CEO was decrying butter as evil as recently as 2010, is now adding butter to their spreads. So better education can help (you may consider this blog our contribution).
Many large food producers, including some fast food chains, have also begun phasing out trans fats from their cooking under pressure from consumer groups. Yet a lot of fast food and processed food continues to be cooked in trans fat. Mostly that’s because the slower rate of oxidation means the oil can be changed less frequently without it going rancid, and the resulting products last longer too. That’s a reason to continue using partially-hydrogenated oils, but not a very good one: other oils have been developed in recent years (by separating out the least-volatile components in plant oils) that last longer and do not contain trans fat.
It’s certainly not, as Carlson speculated, because of the taste. If you’re anything like me, you’ll be fond of quoting the maxim “fat is flavour”. Keep in mind, though, that it’s a gross simplification. In reality the compounds that add flavour are fat-soluble, and so they’re found along with the fat in foods. The fat itself really does have “no particular taste”. (Colbert: “That’s America’s favourite flavour.”) “Fat is a necessary but not sufficient condition for flavour” doesn’t pack quite the same rhetorical punch, but it’s more accurate.
There’s simply no reason for consumers to ever choose to eat trans fats, except because the products they want are not available without them—at least, not at a price they are willing to pay. The looming ban on trans fats in the US should solve the availability issue. And while the poor are the biggest beneficiaries of cheap food, they also bear the brunt of the negative effects. It’s clear at this point that the cost savings of partial hydrogenation are a false economy.
My biggest worry is the extent to which partially hydrogenated fats might be replaced with Palm Oil, which is another in that small handful of saturated plant fats. Large swathes of tropical rainforest are being razed every year to make way for palm oil plantations, which is one of the worst things you could do from a climate change perspective. Even as trans fats are cast on the dust heap of history, we will all have to remain vigilant consumers.
If you liked this post, we have more where this came from and you’ll hear about it when you follow our Twitter feed.
The first edition of our Meal Planning guide, The Seasonal Supper, is available now! Check out the free, one-week Think Spring preview and then sign up to purchase the full four-week Summer Simplified meal plan. Our eating philosophy is food-based, so you’ll have no trouble avoiding trans fats.



Great job Zane!
Zane, thank you so much for this explanation. It really helped me understand. I changed to real butter some years ago and have been trying to avoid palm oil, not only in food but in cosmetics and other places it may be used due to the destruction of rain forests and animal habitats that palm oil plantations have wrought.